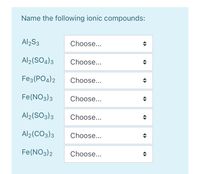
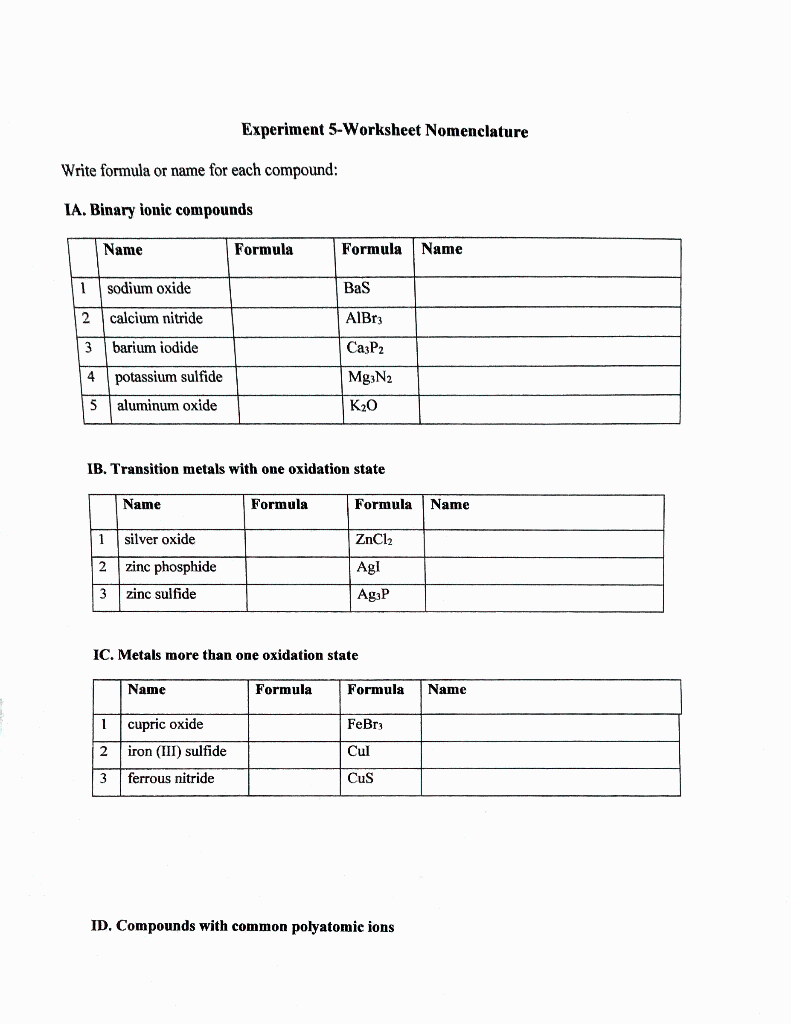
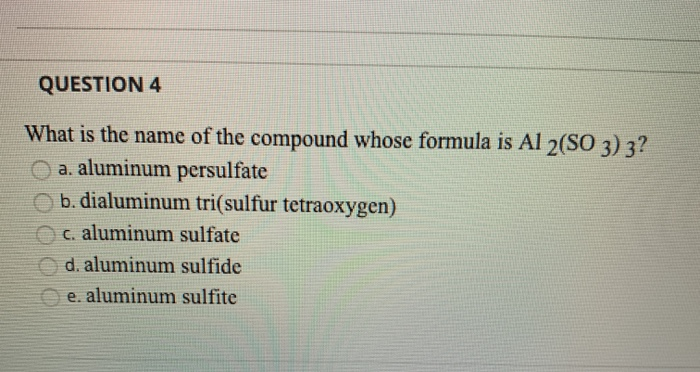
SOLVED: Name each of the following compounds. Assume the acids are dissolved in water. a. HC2H3O2 b. NH4NO2 c. Co2S3 d. ICl e. Pb3(PO4)2 f. KClO3 g. H2SO4 h. Sr3N2 i. Al2(SO3)3
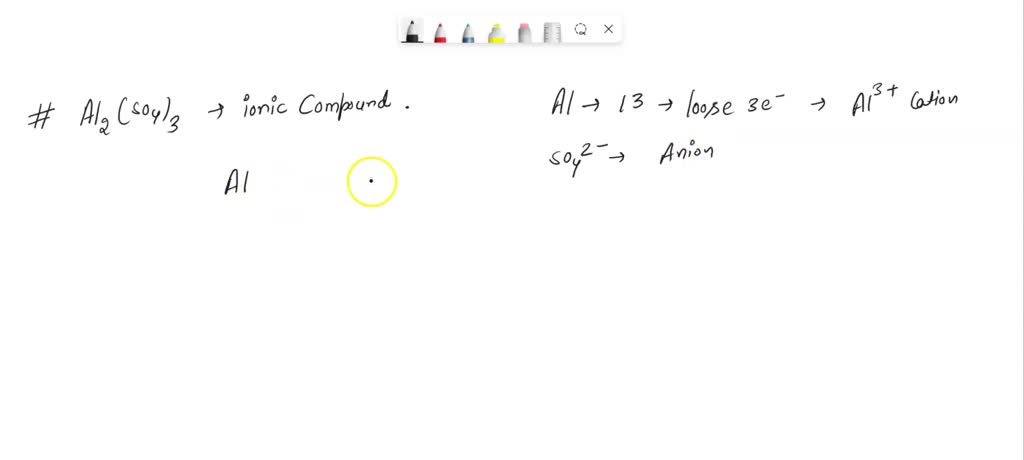
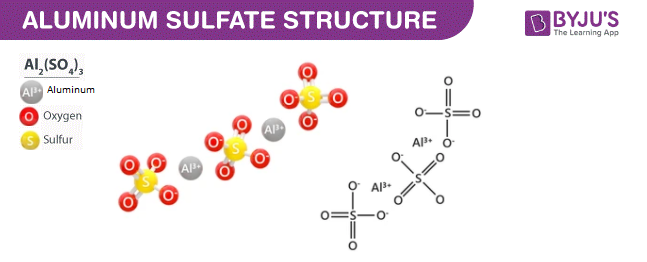
SOLVED: The chemical formula for the ionic compound aluminum sulfite is Al2(SO3)3. Explain why there are 2 cations for every 3 anions in this compound.




.png)